Magnesium hydroxide (Mg(OH)₂) is a workhorse in the world of inorganic flame retardants. Its effectiveness is not due to a single magic trick, but rather a sophisticated, synergistic combination of three distinct mechanisms that create a powerful barrier against fire. It is a master of defense, operating on both physical and chemical fronts.
Mechanism 1: The Cool-Down – Endothermic Decomposition
This is the primary and most crucial mechanism. When the surrounding temperature rises to approximately 340°C, magnesium hydroxide begins to decompose, breaking down into magnesium oxide (MgO) and water vapor (H₂O). This chemical reaction is highly endothermic, meaning it absorbs a significant amount of heat from its immediate environment—about 1.3 kJ per gram.
In a polymer matrix (like plastic or rubber), this powerful cooling effect acts as a "heat sink." It drastically lowers the temperature of the material, delaying its thermal degradation and slowing the production of combustible gases that fuel a fire. It essentially starves the fire of the thermal energy it needs to propagate.
Mechanism 2: The Gas Shield – Dilution and Isolation
As magnesium hydroxidedecomposes, it releases a large volume of water vapor. This non-flammable gas serves a dual purpose:
Dilution: It dilutes the concentration of flammable volatile gases and oxygen present near the surface of the burning material. Since fire requires a specific fuel-to-oxygen ratio, this dilution disrupts the combustion process at a fundamental level.
Inert Barrier: The released steam can also form a temporary inert atmosphere over the material, further isolating it from the surrounding air (oxygen).
Mechanism 3: The Protective Layer – Ceramic Char Formation
The final product of the decomposition is magnesium oxide (MgO), a refractory material known for its high thermal stability. This fine, solid residue forms a coherent, ceramic-like char layer on the material's surface. This layer acts as a robust physical barrier with three key functions:
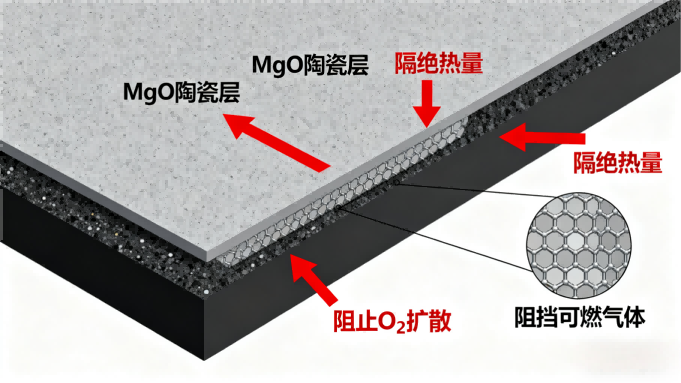
It insulates the underlying virgin material from radiant heat.
It impedes the diffusion of oxygen to the pyrolysis zone.
It hinders the escape of further combustible gases from the polymer.
Furthermore, this protective char helps prevent the dangerous "melt-drip" phenomenon in plastics, which can spread fire to new areas.
Advantages and Applications
Compared to halogen-based flame retardants, magnesium hydroxide is favored for being halogen-free, thus avoiding the production of toxic, corrosive smoke during a fire. It is also non-toxic and environmentally benign. This makes it ideal for applications in wire and cable insulation, building materials, transportation components, and plastics used in electronics, where safety and environmental impact are paramount.

