In today's world where fire incidents occur frequently, flame-retardant materials have become an indispensable line of defense for protecting life and property. Among the many flame retardants, magnesium hydroxide stands out with its unique combination of properties, hailed as the "Green Guardian" in the field of flame retardancy. It not only effectively inhibits flame spread but also achieves a precious balance between environmental protection and safety, making it a highly favored choice for flame retardancy in polymer materials.
I. Excellent Flame Retardancy and Smoke Suppression Mechanism
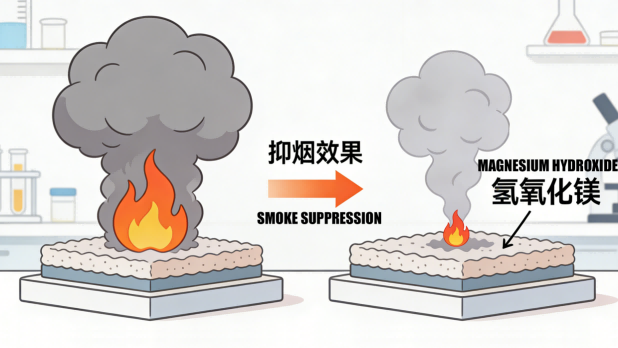
The flame-retardant action of magnesium hydroxide stems from its precise chemical design and physical properties. When the temperature rises to approximately 340°C, magnesium hydroxide begins to decompose into magnesium oxide and water vapor. This endothermic reaction effectively lowers the surface temperature of the material, delaying the thermal decomposition process. Simultaneously, the released water vapor dilutes the concentration of oxygen and combustible gases, and forms a dense magnesium oxide coating on the material's surface, isolating the transfer of heat and oxygen. This triple synergistic mechanism of "cooling, dilution, and coverage" endows magnesium hydroxide with efficient and reliable flame-retardant performance.
It is particularly noteworthy that magnesium hydroxide performs excellently in smoke suppression. Deadly smoke during fires is often a major cause of casualties. The decomposition products of magnesium hydroxide are non-toxic themselves and can adsorb some of the smoke particles generated during combustion, significantly reducing smoke density and buying valuable time for evacuation and fire rescue.
II. Outstanding Environmental and Safety Advantages
Compared to traditional halogen-based flame retardants, the core advantage of magnesium hydroxide lies in its excellent environmental friendliness. It does not produce toxic or harmful substances during production, use, or disposal. Its decomposition products are only magnesium oxide and water, unlike halogen-based flame retardants which release hazardous substances such as corrosive and environmentally persistent dioxins and hydrogen halides. This makes materials containing magnesium hydroxide easier to handle at the end of their life cycle, and even allows for recovery and reuse through appropriate processes, aligning well with the principles of a circular economy.
In terms of biosafety, magnesium hydroxide itself has very low toxicity and is friendly to humans and the environment. Its decomposition does not release irritating or toxic gases, significantly enhancing safety at fire scenes. Additionally, magnesium hydroxide possesses anti-acid properties, neutralizing acidic gases produced during material combustion, further reducing secondary hazards.
III. Broad Applications and Synergistic Enhancement Technologies
Leveraging these advantages, magnesium hydroxide is widely used in polymer materials such as polypropylene (PP), polyethylene (PE), polyvinyl chloride (PVC), epoxy resins, rubber, and coatings. It is especially suitable for applications with high requirements for environmental protection, smoke suppression, and electrical performance, such as wires and cables, building panels, housings for electronic and electrical appliances, and interior materials for transportation vehicles.

However, magnesium hydroxide also faces challenges, such as the relatively high loading required (typically 50-60% to achieve good flame retardancy), which can affect the mechanical properties of the base material. To address this, researchers employ technologies like nano-sizing, surface modification (e.g., using silane or titanate coupling agents), and synergistic compounding with other flame retardants (e.g., phosphorus-based compounds, expanded graphite). These techniques significantly improve its dispersion and compatibility within polymers, achieving an optimal balance between high-efficiency flame retardancy and mechanical performance.
Conclusion
As a representative inorganic flame retardant, magnesium hydroxide plays a crucial role in modern fire safety with its comprehensive characteristics of high efficiency, low smoke and toxicity, and environmental friendliness. As global environmental regulations become increasingly stringent and public awareness of safety continues to grow, the application prospects for magnesium hydroxide flame retardants will broaden even further. Future research and development will continue to focus on overcoming its current limitations through technological innovation, further unlocking its potential as a "Green Guardian," and providing a solid material foundation for building a safer and more sustainable societal environment. On the path towards harmonious coexistence between technology and nature, the development of environmentally friendly flame retardants like magnesium hydroxide undoubtedly represents a significant step forward.

